Research - (2022) Volume 13, Issue 3
Received: 04-Mar-2022, Manuscript No. CSJ-22-23430;
Editor assigned: 18-Mar-2022, Pre QC No. P-23430;
Reviewed: 21-Mar-2022, QC No. Q-23430;
Revised: 22-Mar-2022, Manuscript No. R-23430;
Published:
31-Mar-2022
, DOI: 10.37421/csj.2022.13.284
Citation: Hussain, Umm-e-Habiba Munawar. “Synthesized, Combined, and Characterized Gelatin Nanoparticles and Collagen used for Antimicrobial Activity.” Chem Sci J 13 (2022): 284.
Copyright: © 2022 Hussain UHM. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Gelatin is a flavorless solid substance along with translucent colorless and brittle than present in dry form. Gelatin (mixture of peptide) is produced by hydrolysis of collagen and is an important part of extracellular matrix and provides mechanical protection to the tissues. Collagen is extracted from the different parts of animal such as skin, bones and connective tissues and also is extracted from different animals like cattle, chicken, pigs and fishes. Collagen is a triple helix molecule comprised of three Alpha helix of glycine and X-Y in which X and Y is mostly Proline and hydroxyproline, respectively. Collagen is rapidly used in cosmetics, pharmaceuticals, tissues engineering construction and most mostly used in dressing system. Gelatin which is obtained from hydrolysis is of two types, type of gelatin ‘a’ and type of gelatin ’b’ with isoelectric point 7 to 9 and 4 to 5 respectively is obtained from acid pretreatment and basic pretreatment. Gelatin nanoparticles are important in biomedical field that instigate the development of new nanoparticles of different synthetic Polymers like that poly ethylene glycol, poly glycolic acid, poly lactic acid, poly alkyl cyanoacrylate and natural polymers(a chitosan, gelatin and dextran) are used for the development of nanoparticles for biological application and drug delivery system. Among all, gelatin nanoparticles are mostly used because of inexpensive, non-toxic, bioactive and biodegradable properties. In gelatin, type-B shows good potential for drug delivery as compared to the gelatin type A. Gelatin based material must be cross linked with glutaldehyde.
Gelatin • Collagen • Chitosan • Glutaraldehyde • Nanoparticles • Scaffold
In current years there has been an increasing interest for using biodegradable materials in packaging, medicine, agriculture, food. A great variety of blend by using the biopolymer is a substitute of presently used of synthetic polymer material. Most common and probable biopolymer are starches, alginates, PLAs, PHA, gelatin and chitosan [1]. Among these, gelatin has increased global demand as it is an important functionally active biopolymer. Gelatins the naturally happening macromolecule and soluble proteins are fashioned by precise fractional hydrolysis of the collagen that is produced from skin, connective tissue and bone of the animals which constitute the amino acid residue by various proportion and combination [2,3]. Gelatin derived from Latin word gelatus have meaning firm or solid was a glowing or translucent, colorless, brittle and flavourless hard material. The house-hold gelatin comes in the arranged form of sheet, granule and powder. Gelatin is a poly-ampholyte compound in nature as it contain both cationic and anionic group. Gelatin polypeptides are composed of many repeating triplet of the alanine, proline besides glycine residues responsible used for distinctive triple helix structure of the gelatin [4,5].
Formation of triple helical structure is completed by two steps, nucleation and the wrapping. The Nucleation step includes positive or optimistic activation energies in addition to it is considerable slow procedure than that of wrapping step where negative or adverse energy was related with wrapping step. Slow nucleation procedure is a rate limiting step thus this process governs the reactions order [6].
Gelatin from terrestrial animal sources was chosen over aquatic source because of the superior strength of gel, melting points and the viscosity. Nevertheless, gelatin of fish capture growing consideration as a substitute to the terrestrial animals gelatin owing to religious constraint and the fitness issues [7]. Colour and clarity both for gel gelatins are vital appealing properties depending at applications used for future. The gelatin colour depends on uses of raw material, and process of the extraction.
Gelatin the soluble protein got by incomplete hydrolysis of collagen the chief fibers protein constituents in the bones, cartilage and skins. Hence the sources, ages of animals, and types of collagens were the entire internal factor inducing the properties for gelatin. Most plentiful sources for gelatins are the pig skins (45%), bovine hides (28.4%), pork and cattles bone (23.1%). Gelatin of fish accounts for low than 1.4% of the total gelatins manufacture in 2007, nevertheless these percentages were double than that for the market data of 2002, signifying that production of gelatin from substitute non-mammalian specie had full-grown in position [8].
The yield of gelatin obtained is calculated by:
% Yield (wet weight basis) = Gelatin’s dry weight/skin’s wet weight of × 100
The two foremost types of gelatins are type-A with iso-ionic points of 7.1 to 9.1 was derived entirely by using acids pretreatments and type-B with iso-ionic points of 4.1 to 5.1, was a result of alkaline pre-treatment. Types of gelatins (type A or type B) obtained is governed through the procedure of collagens hydrolysis. Type-A and type-B have different isoelectronic points [9]. Type-A has isoelectric point 7–9 despite the fact that type-B has isoelectric point 4–5. Every types of gelatin have diverse drugs release potentials for various kind of nanoparticles. It was revealed that type-B GNPs shows best potential drugs deliveries than the type-A [10]. Furthermore, gelatins of type-B really capture the DNA molecules accordingly increasing the trans-faction efficacy of the carrier. Gelatin that is commercially available has heterogeneous mixtures of polypeptide, existing in variable molecular weight of various ranges that is of thousands to millions Dalton [11].
Gelatin the naturally multi-purpose biopolymer has different vital application owing to the lower cost, calm availability, biocompatible or biodegradable nature in addition to presence of plentiful active groups [12]. Owing to the favorable assets like highest water solubility, nontoxicity and thermo-reversible sol gel transitions, highest mechanical strengths and resistance in the dry states, moisture caused by binding sufficiently amount of water, gelatin are far used as raw material in photography, pharmaceutical, food industry and cosmetic industries. It is used as H2O soluble capsule with coatings material for oral drug, stabilizers of photosensitive reagent in photographic film, adsorbent of diluted chemical and adhesives agent [13,14].
Gelatin is the commonly used gelling agent in cosmetic manufacturing, foods, pharmaceutical and photography’s. The materials containing the gelatin are called the gelatinous substances. Gelatin an irreversibly hydrolyzed forms of collagens is used as foodstuffs. Gelatin is found in many gummies candies along with many other products like marshmallow, and gelatin desserts, ice cream and yogurts. In food industry, gelatin the water soluble biopolymers is used as great ingredient to advance the elasticity, stability and consistency of foods. They are utilized in confectionary (principally to provide texture, foam stabilization and chewiness), low-fats spread (for providing the reduction of fat and creaminess), in dairies (for providing the stabilizations and texturizations), baked goods (to provide emulsification, gelling and stabilization) and in meat product (for providing the water-and fat- bindings) [15].
Gelatin nanoparticles
GNPs are biodegradable, inexpensive, nontoxic and bioactive compounds are very hopeful conditions for drugs deliveries and the skillful drugs release. Various mechanical assets of gelatin nanoparticle size such as thermal ranges and swelling behaviors are dependent on amphe-tatic interaction of gelatin. Dissimilar gelatin nanoparticles are prepared through manipulating the various physical and chemical properties of gelatins [16,17]. Gelatin-based material needs to be crosslinks with the glutaraldehyde or many other bifunctional cross-linker like N- hydroxy succinimide, genipin, and transglutaminase and aldehyde group to reduce the insolubilities at higher temperatures, reducing the swellings in the water and later drugs release from nanoparticles. The drug release capacity was proposed depend on the cross-linking’s densities of gelatins [18]. The gelatin nanoparticles are produced by diverse technique together with de-solvation, coacervation separations, nano precipitation, emulsifications, reverse-phase emulsion, and solvent evaporation.
Two-step de-solvation method
In this, a de-solvating manager is additional to the aqueous solution of gelatin to dehydrate the molecule of gelatin. Lower molecule mass gelatin is rejected and residual higher molecule mass portions are liquified in liquid or water and after that ethanol is added in the solution drop-wise at the controlled PH. Crosslinker is supplement and rotate or stirred it at 601 rpms at 41°C intended for the 12 hours to get the hardest, uniform and spherical nanoparticles [19]. Purification is done by acetone: water afterward wide centrifugation and lastly lyophilization.
Simple coacervation method
Simple coacervation method was used for preparing the even and small sized particle [20]. After the liquid- liquid separation of phase, polymers settles down in solutions creating the two visible and immiscible phase. Natural salt or alcohols are usually used to get nanoparticles. Though, oppositely charging macromolecule like proteins are proposed the coacervation that is also called the complexed coacervation. Dryness of gelatin finally result the formation of gelatin nanoparticles that are used for crosslinking with another cross-linked agent like glutaraldehyde [21,22].
Solvent evaporation method
Solvent evaporation method in which the single emulsion and double emulsion used for creation of gelatin nanoparticles. Gelatins and drug in the aqueous phases were homogenized by the oil phase and at that point cross links with glutaraldehyde [23]. After that solvent was evaporated. Solidified nanoparticles were collected and wash away through the distill water for removing the additive for example surfactant, and at that moment lyophilized [24].
Micro or mini emulsion
In this technique, gelatin solutions are discharged into the surfactant solutions in the hexane, cross-links with glutaraldehyde then lastly hexane is dispersed and gelatin nanoparticles are improved [25]. Micro emulsion may control the sizes of nanoparticles and thus it considered as advantageous.
Nano-precipitation or solvent dis-placement technique
In nano precipitation water was used by the way of solvent point or phase and in water gelatins and drugs molecule was dissolved. Aqueous phase was supplement to the ethanol as an non-solvent phases comprising the poloxamer as stabilizer. Gelatin nanoparticles were designed at junction of water and ethanol are later cross- links by using the glutaraldehyde [26].
Collagen
Collagen the greatest plentiful protein is the representative of closely 31% of the total protein in the body of animal. Collagen the main constituent of the extracellular matrices is important for protections of the tissue, organ and the physiological regulations of the cellular environments [27]. Unique physical properties of fibers of collagen discuss the mechanical integrity for the skins forming the thick networks throughout the epidermis. Main functions for the networks are for providing the mechanical supports to dermis. In count of collagens and elastins collectively constitute the extracellular matrices that give to skin its structural elasticity or firmness [28]. Collagen the animal protein is present in the connective tissue of animals. Collagen typically originate from the fibers tissues like tendon and ligament, and similarly is plentiful in cornea, cartilages, bone, intervertebral discs, gut, and blood vessels. Insoluble collagen is main fibrous protein originate from the extra-cellular medium of the skins collectively with the elastins and the hyaluronic acids. Family of collagen contains the 28.1 various protein [29,30], that accounts the 25.1% - 35.1% of the total proteins weight in the mammal and show the vital role within the structures of different tissues for example skins and bone to provide the firmness and integrity [31,32]. Collagen of type-I is desolved or liquified in the acetic acid to create a solution. Hydrolyzed collagens contains the small peptide with lower molecule mass about 0.4 - 8 k-Da, formed from the natural collagens. Owing to the lower molecule mass, hydrolyzed collagens are easily edible, distributed and absorbed in human beings. Owing to lower molecule mass, hydrolyzed collagens have numerous advantages. As compare to the natural collagens hydrolyzed collagens are extremely digestible and hydrolyzed collagens are simple to the absorbent and dispersed in human beings’ bodies. The collagen peptides have the great influence at mass loss and are active at keeping the mass with no harm effect [33] (Figure 1).
Presently, the main source of collagens type-I is bovines and porcine epidermis. Right now, there were much report on separation and characterization for collagens obtained from fish waste of fresh water. There were very fishes of fresh water especially of a Carps family. Cyprinidae are originating from freshwater lake and river around from Southwest Asia and Southwest and East West Asia [34] besides expended worldwide. Current study focused on separation and characterization of collagens obtained from the scale of fishes. The collagen is secluded from scale of fish and the solidified tissues by the treatment of demineralized acetic acid.
Founded happening the structure beside 3D body collagen grouped into fibril forming collagens, fibril-related network-forming collagens, trans membrane collagens, connected collagens, basement membrane collagens, anchoring fibril beside numerous others by means of restricted functions [35,36]. Even though twenty-seven diverse class of collagen encompass be recognized, collagen of type I be mainly extensively happening collagen into the connective hankie. In human body 80 to 91% of the intact collagen consists of the fibril forming collagens. Type-2 beside type-6 collagen fibrils is concerned into configuration of the fibrillar impression into articular cartilage [36,37]. IVType collagen into distinction form a two-dimensional reticulum beside the supplementary bendable triple twirl was constrained in the direction of cellar membrane [36,38]. Collagen be chiefly produced by fibroblasts into joined tissues except abundant epithelial cell create dense collagens types [39]. The diverse collagen beside structure they prearranged has the determination to help tissue-resist stretching.
Molecules of interstitial collagen are tranquil of 3 a-chains lay to resttwine into the triple helix of collagen. This particular configuration frequently stabilize through the inter and intra manacles hydrogen bond, be a creation of an practically incessant revealingly of the Gly-X-Y- succession into which X be usually proline beside Y be usually hydroxy proline. Merely an extremely petite N- beside C- terminal region be known as telopeptides, which accomplish not prearranged the triple-twisted structure into put off they be mainly comprise through lysine in addition of hydroxylysine remains into adding to their derivatives of aldehyde into both the inter beside intra -molecular covalent cross-connection.4 to 8 molecules of collagen into the fractious-segment be stabilize beside strengthen through the covalent bond on the way to generate the fundamental unit of the fibrils of collagen. Consequently, the physically powerful or firm environment of pelt, bones and tendons is for the reason that of the fundamental structure intended by numerous of these collagens fibrils are crosslinked. The secluded and cleaned collagens were characterized by the FTIR, amino acid analysis and SDS page analysis. Temperature of denaturation for collagens was valued via CD or circular dichroisms spectroscopy at diverse temperature and induced changes by thermal in viscosities.
The utilization of swiftly collagens are growing up in makeup and in the industry of pharmaceutical. Amongst numerous kinds, I-type collagen had widely been utilized for wound dressing system and development of tissue engineering as a biomaterial because of less antigenic and highly adhesion properties of direct cell [40]. Chief collagen applications are as delivery of drug system are membranes of collagen for delivery which is transdermal, ophthalmology shields, collagen sponges, tablets within dentistry, NPs for delivery of gene in addition to substitutes of bone [41]. Composite scaffolds hydroxyl-apatite with collagen for bone tissue engineer is a very active ground because the hydroxyl-apatite and the collagen are key component in natural bones. Chitosan is an amino polysaccharide is a derivative of chitin obtained by de-acetylation is widely useful in biomedical fields like wound dressings besides the drugs delivery system. The bio-degradable and bio-compatible chitosan is added to scaffolds and porous collagen-based scaffolds of bone, strengthened by chitosan fibers with suitable properties of porosity is ready [42]. Collagen has become the biomaterial for various important medical applications including the collagen has a good safety profile and understanding of collagen role in metabolism, catabolism, wound healing, and interaction between cells and collagen has been greatly improved [43]. The sponges of collagen is an additional advantages intended for soften repairing of tissue cause of its strength of wet permit fastening the material providing a prototype intended for the growth of tissue [44].
Bio-active properties of gelatin and collagen
The recent knowledge on the anti-microbial hydrolysates properties and obtained peptide via gelatin and collagen are extremely rare. Antimicrobial activities of peptides fractions get from the tuna and the squid skin gelatin between the range of 1 to 10 k-Da and <1 k-Da. Hydrolysates utilizing the agar-diffusion tested against seventeen bacteria stains (for Gram negative and positive) the acidophilus Lactobacillus and the animals Bifido bacterium. The lattices, Shewanella putrefaciens in addition to the phosphoreum are photo bacterium establish to be mainly species which are susceptible. Compact weight of molecules in the fraction of peptide that were connected to the masses removal, amino acid better exposure residue in addition to charges, along with acquisition of structure, are recommended as facilitating factors the interacted to the membranes of bacteria [45].
The gelatin and collagen peptides which are anti-oxidative can protect free radical against living cell facilitated damage oxidative. Consequently the scavenging species of free radical is peptides of antioxidant by central mechanism boost oxidation against viability of cell and ultimately death of cell occur [46]. Anti-hypertensive peptides were molecules of peptide that might lesser the blood pressure that is consumed via inhibition vaso-active enzyme like as enzyme converting angiotensin. ACE participate a fundamental role in blood pressure regulation through angiotensin-rennin system in addition to the enzyme inhibition is measured to helpful approach which is therapeutic in hypertension treatment [47,48].
Applications
The cosmetic and conventional victuals, pharmaceutical, photographic purpose of gelatin be base mostly scheduled its forming of gel in addition to visco-elastic property. In recent times, particularly into the victuals manufacturing, an escalating numeral of novel application contain be establish intended for gelatin into yield like as emulsifiers, colloid stabilizers, foaming agents, biodegradable packaging materials, fining agents in addition to microencapsulate agent, into procession by means of the mounting tendency to restore mock agent by means of supplementary ordinary ones. Furthermore, into numerous belongings, these studies be devoted to by means of collagens beside gelatins beginning another source in the direction of terrain-base animals.
Applications of GNPs in nano-biotechnology and biomedical sciences
Usage of the nanotechnology besides science of life deals innovative opportunities to diagnose the medical measures. The nonmaterial take energetically recycled in diverse applications resembling the drug deliveries system, cancer therapy, scaffolding of tissues, diagnostics, and tissues engineering techniques and healing in medication [49,50]. Strong relationship subsists among its carrier system and drug, thus permitting inflection of the bio-distributions of drug and pharmacokinetic assets. Nanoparticles remain maximum examined among diverse drug delivery systems and drug carriers. Targeted drug delivery and Controlled are eagerly recycled in traces to improve localization of medications to specific cells and tissues, lessening their potential toxicity and collaboration consequently growing the healing guide of surviving drugs. Receptors-mediating painkiller directing was unique for favorable, safe or effective methodology recycled in drug distribution scheme precisely to its spot of act [51].
In pharmaceutical productions utmost of the effective anticancer medicines posture a chief problematic of pitiable solubility laterally with additional adverse pharmaceutical properties and physicochemical, which are chief restrictions in their experimental direction [52]. The efficiency of medicine treatments is predictable to be significantly developed by measured drug deliveries schemes. Gelatin Nanoparticles are some utmost appropriate applicants within dropping poisonous matter related with utmost of the medicines and might be recycled as a favorable applicant for measured drug issue [53].
Crossing the blood brain barrier
Barrier of blood brain with difficult physical building have vital central nervous system which hinders permitted dispersal for the circulating molecule since plasma within head. Since for that obstacle through whole central nervous system popular for mind syndromes persist inoperable. Nano-particulate classification has the aptitude to stun this wall [54]. The medicines managed complete intravenous vaccination can be conveyed transversely BBB with the aid of NPs medicine supply organization more professionally. Medicines comparable to endorphin, kyotorphin, dipeptide loperamide, tubocurarine and doxorubicin, might be overloaded against these nano-protein elements [55]. NPs consume the capability to simply movement over vessels and can enter their barriers to extent their locate of act owing to their perfect surface functionalization and minor extent [56,57]. Inactivation through short breathing, Si-RNAs remain the newly exposed progressive method recycled in genetic factor silencing. Conversely, this system absences the wanted diffusion of slight RNA into the film [58,59]. GNPs deliver a well-organized vehicle provide safety against degradation for carriage of these RNAs toward the chamber and also have safe degradation yields [60].
GNPs as DNA carrier
GNPs devour been considered by numerous investigators as ship for gene carriage [61]. Gene appearance in mice consequent to intramuscular inoculation of DNA-gelatin Nano scopes remained superior and further lengthy associated to an equivalent quantity of DNA complexed or bare DNA by lipofectamine [62]. Improved gelatin nanoparticles remained recycled in mortal breasts cancers chambers for intra-cellular concentration. Additional bioactive hybrid mixtures similar siloxane fusion with gelatin remained also described as harmless innovative non-viral vector and efficient for gene distribution [63,64].
GNPs as bactericidal bodies
The silvery NPs filled with gelatin hydro gel packs were too initiate to be operative in removal of P. aeruginosa and E. coli approximately 101% reserve proportion. Then the hydro gel consumes lethal possessions at usual skins. Conversely, gelatins hydro gel-controlled silver nitrate at minor applications (1.1 weight %) are measured towards the additional harmless towards the covering chambers [65]. Correspondingly, gelatin-based sponges combined by silver nanoparticles were organized through resolution plasmas procedure. This is exposed that AgNP/gelatins elucidations besides scrapers showed outstanding bactericides action in contradiction of two bacteria’s that is S. aureus and Escherichia coli. Gelatin NPs remained too recommended by way of immune-logically adjuvants to trigger besides enhancement immunities beside antigens, either humoural or cellular [66,67].
The roles and potentials of gelatins in tissues engineering’s
Tissues manufacturing are some of the greatest stimulating turfs of nanotechnology. Tissues manufacturing emphases at cohort vitro of bio compatibles tissue. Usage of nano composites supports tissues manufacturing have involved comprehensive inquiry attention in new centuries owing to their biological source [68]. Currently numerous kinds of nano composites are strained in tissue manufacturing approaches, to mark absorbent frameworks. Nano composites frameworks are recycled within renovation of tissue for example bone, muscle, tendon, ligaments, cartilages, tissues interfaces and another multi-tissues organ [69]. Composite schemes synergistically chain two or extra supplies in instruction to yield an innovative scheme with novel possessions. Gelatin built composite schemes deliver new selections for tissues manufacturing. Gelatin a denatured biopolymer is the collection for gelatin framework measure able may evade distresses for pathogen transmission in addition to immunogenicity connected with collagens [70]. Maximum effort to build the usage of the gelatins nano composites emphasis within jawbone faults. Wounds infection and bones non-union were two greatest renowned challenges for bones defects. The productions and applications of gelatinsbuilt ceramic for medicine transfer besides bones re-development were the dynamic development. Gelatin nanocomposite are able to be designed with diverse sorts of polymers, together synthetic and natural, permitting for determination of the usage. Insulin similar growths factor-1 is testified for growing the 5 periods surge within amount of chambers, once cured through the act ionized gelatin nanoparticles covering the growth factor-1 plasmid associated by unchanged particle [71].
Experimentation
Synthesis of GNPs (gelatin nanoparticles): GNPs are synthesized by the two-step de-solvation process. Take 1.16 gram gelatin and resolved in 26 mille-litter water at steady heating 25 ml ethanol is added in gelatin solution which acts as de-solvating reagent to ppt the higher molecular mass of gelatins (HMV). HMV is re- resolved in 25 ml distill water and stirred it at 601 rpm at steady heating. PH is in tune between 2 and 12 ml and ethanol is added drop-wise to shape the Nanoparticles. At last add 250ul glutaldehyde as crosslinked agent along with stirring it at 601 pm in favor of 12 hours.
Synthesis of scaffolding: Scaffolding is prepared via blended scheme. The chitosan along with collagen was dissolve in 0.6 molar acetic acid solutions and gelatin discrete in demineralized water with reflex stirrer in addition to kept i t at 5°C. Separate solution was blend together to get the one percent concentrations of every polymer within mixture. Blend is centrifuged on 2401 × g for 31 mint to remove the air. Resultant mixtures are loaded in cylindrical mould along with kept it at 11°C for 21 hours and then freeze up dried it at -21°C. Obtained scaffolds were store at 4°C in tightly close container.
Antimicrobial activity: The Nutrient Agar and Potato Dextrose were used to prepare sterile 9cm petri plates, and these plates were stored at 4°C, until they were required. The microbial starter cultures were prepared 24 hours before and incubated at 25°C. The selected compound was used to make concentrated extracted in distilled water. The extracts were not heated to sterilize prior to their use due to the possibility of affecting the active ingredients of compounds. The Nutrient Broth solution and the selected microbial species culture grow at 25°C. The 4mm paper discs (sterilized) were infused with the fresh compound extracts and were placed on the newly seeded fungal. Following application of the neutraceutical disks to each plate, all plates were incubated at 25°C. Following the completion of incubation, the zones of inhibition were measured. The inhibition zone was measured with help of the scale in millimetres, after every 24 hours up to a total of 96 hours. The results of the inhibition zone in respect to the antimicrobial activity of the synthesized scaffold.
Nanoparticles size of gelatin
Nanoparticles have the range of size between 100-300 nm. The given Nanoparticles of gelatin have hydrodynamic diameter 243.44 and poly dispersity index 25.14%. The first peak has intensity of 357.9 nm and second peak intensity is 40.63 nm.
Result of chitosan based collagen and gelatin scaffold composite
Porosity of scaffold: The porosity of scaffold is determined by ethanol infiltration method. The weighed scaffold (Wd) is immersed in ethanol (absolute) for infusion in scaffold for 15 minutes at room temperature at reduced pressure in desiccator for removing the air bubbles. After this scaffold material is detached and evaluated [Ww] with ethanol at surface. The porosities (%) is considered by the given formula.
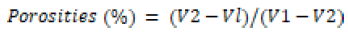
The V2 shows volumes for ethanol beforehand and V1 shows volumes for ethanol after infusion of scaffolding. Vl signify volumes of alcohol afterward the removal of scaffold samples from the liquid (Tables 1 and 2).
| Composition | Biopolymers | Polymer Composition in Dispersion (% W/W) | Solutions |
|---|---|---|---|
| 20% GEL, 40% COL and 40% chitosan |
GEL, COL and chitosan | 0.4%, 0.8%, 0.8% | 0.5M acetic acid, water |
| Composition of Scaffold | Porosity |
|---|---|
| Col/Gel/chitosan | 62% (-,+) 3 |
FTIR studies of scaffold of col: Gel
The synthesized scaffold of gelatin and collagen were also characterized by using FTIR scan. To obtain Infra-red spectrum of synthesized scaffold Carry 630 FTIR spectrophotometer, present in Chemistry Department of UET, Lahore was used. They range available on this spectrophotometer was 650- 4000 cm-1. The C-H give peak at 1155.5 cm-1. The C-N stretching of primary amines give the peak at the range of 1250-1020cm-1 and gelatin collagen scaffold give the peak at 1252 cm-1. The CO-N group give the peak at the range of 1398-1266 cm-1 of IR and scaffold give the peak at 1394 cm-1. The C=N group of secondary amine give the peak at the range of 1690-1640 cm-1 and scaffold give the peak at range of 1632 cm-1. The C=O functional group give peak at 2117 cm-1. The N-H stretching of secondary amine give peak at 3300-3310 cm-1 and obtained peak is at the range of 3267 cm-1. The OH stretching give peak at 3550-3200 cm-1 and obtained peak at the range of 2981 cm-1 (Figures 2 and 3) (Tables 3-5).
| S.No | Test Compound | 24 hours | 48 hours | 72 hours | 96 hours | Remarks |
|---|---|---|---|---|---|---|
| 1 | Col: gel: chitosan scaffold | 12.4 | 12.3 | 12.2 | 12.2 | Susceptible |
| S.No | Test Compound | 24 hours | 48 hours | 72 hours | 96 hours | Remarks |
|---|---|---|---|---|---|---|
| 1 | Col: gel: chitosan scaffold | 24.7 | 24.6 | 24.4 | 24.2 | Susceptible |
| S.No | Test Compound | 24 hours | 48 hours | 72 hours | 96 hours | Remarks |
|---|---|---|---|---|---|---|
| 1 | Col: gel: chitosan scaffold | 11.9 | 11.9 | 10.7 | 10.7 | Susceptible |
In the present project an attempt was made to synthesize the gelatin nano particles by two-step de-solvation method and after that synthesize the collagen-based gelatin scaffold and in this the collagen is mixed with chitosan to improve the antimicrobial activity of scaffold. The particles size of gelatin nanoparticles (GNPs) is analyzed by particle size analyser and porosity of the scaffold is determined by the formula. The collagen-based gelatin scaffold is characterized by FT-IR and the IR studies conducted showed that there was not much significant change observes in the IR peaks of scaffold, and the porosity size shows the correctness of scaffolding. The study on the antimicrobial activity of the scaffold revealed that the antimicrobial activity of the gelatin scaffold was enhanced due to collagen and chitosan as compared to the activity of gelatin scaffold.
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Google Scholar, Crossref, Indexed at
Chemical Sciences Journal received 912 citations as per Google Scholar report