Measles virus (MeV) structure, mechanism of infection, symptoms, diagnostics, prevention and treatment
Tel: +1-847-682-0655, Email: bioinnovastion04@yahoo.com
Abstract
Measles Virus (MeV) Morbillivirus hominis belongs to Paramyxoviridae family. It is enveloped virus with single-stranded, negative-sense RNA. Measles virus infection is mostly seen in the winter and spring. The virus spread through direct contact with discharge from infected person nose, or throat. Symptoms are usually within 7 to 14 days from infection initiation, and include high fever, cough, runny nose (coryza), and red/ watery eyes. After 3 to 4 days from these initiated symptoms a tiny Koplik spots (white spots) appeared inside the mouth followed by rashes on the face that spread to cover all patient body causes patient temperature rise for more than 104-degree Fahrenheit (40°C). Common complications from measles are ear infections, and diarrhea that are usually occur for about 10 % of patients.
Measles illness can be serious for younger children, pregnant women, and people with weak immune response with symptoms like Pneumonia as second bacterial infection, and Encephalitis (serious infection of the brain) that need hospitalization or the patient could die. Some patients suffer from late longterm complications such as a rare, progressive neurological disease known by the name Subacute Sclerosing Panencephalitis (SSPE), a rare and serious neurodegeneration disorder that could cause from mutated measles virus infection infect patient central nervous system. Measles disease was eliminated from United State in the year 2000, but due to unvaccinated tourists, and vaccination declines in United States, in the year 2025 measles illnesses is back. The comeback measles in United States caused the attention to this virus mechanism of infection, and symptoms in order to develop more efficient immunization and treatments.
Keywords
Morbillivirus hominis • Measles symptoms • Koplik spots • Encephalitis • Subacute Sclerosing Panencephalitis (SSPE) • MMR • MMRV • Real-time Reverse Transcription-Polymerase Chain Reaction (rRT-PCR) • Enzyme Linked Immune Assay (ELISA)
Introduction
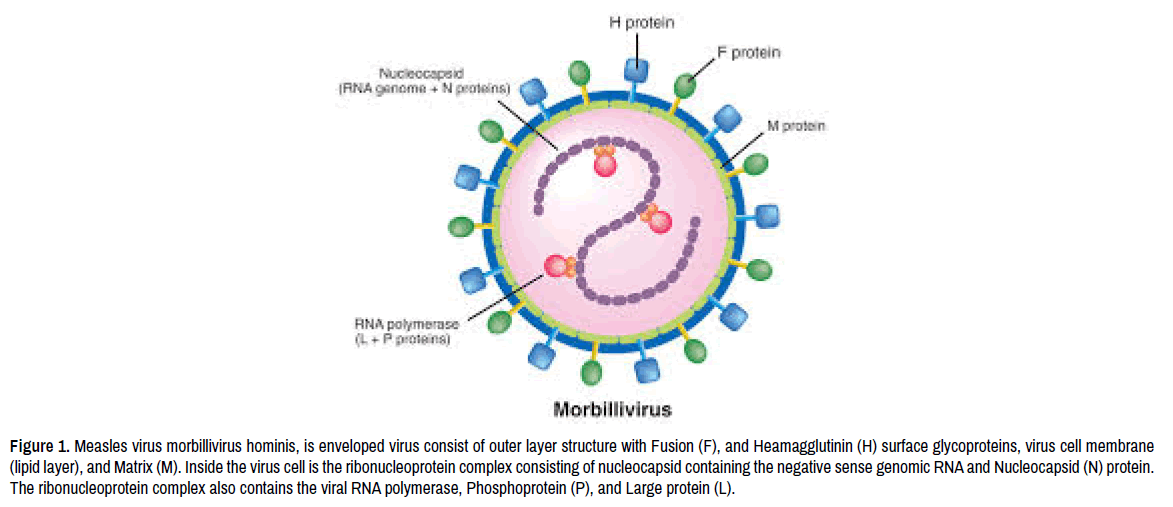
Measles virus morphology (Figure 1) is an enveloped virus with a negative-sense genomic RNA. The Envelop is the virus outer layer with two surface glycoproteins Hemagglutinin (H) and Fusion (F). These two-surface glycoproteins assist the virus to attach and inter the host cell [1]. Under the virus outer layer is the matrix protein connecting the virus envelop to the virus core Ribonucleocapsid (RNP) [2]. The virus nucleocapsid (N) is helical structure contain tightly the virus negative-sense RNA to encapsulate the virus RNA. The virus nucleocapsid also contains Phosphoprotein (P), and Large (L) Protein, both forming the enzyme RNA polymerase complex with function to replicate the virus RNA inside host cell [3]. Other virus proteins are C and V proteins; both are virulence factors with function to suppress the host's immune response allowing the virus to replicate and spread more effectively inside host cells [4].
Figure 1. Measles virus morbillivirus hominis, is enveloped virus consist of outer layer structure with Fusion (F), and Heamagglutinin (H) surface glycoproteins, virus cell membrane (lipid layer), and Matrix (M). Inside the virus cell is the ribonucleoprotein complex consisting of nucleocapsid containing the negative sense genomic RNA and Nucleocapsid (N) protein. The ribonucleoprotein complex also contains the viral RNA polymerase, Phosphoprotein (P), and Large protein (L).
Measles Virus (MeV) mechanism of infection
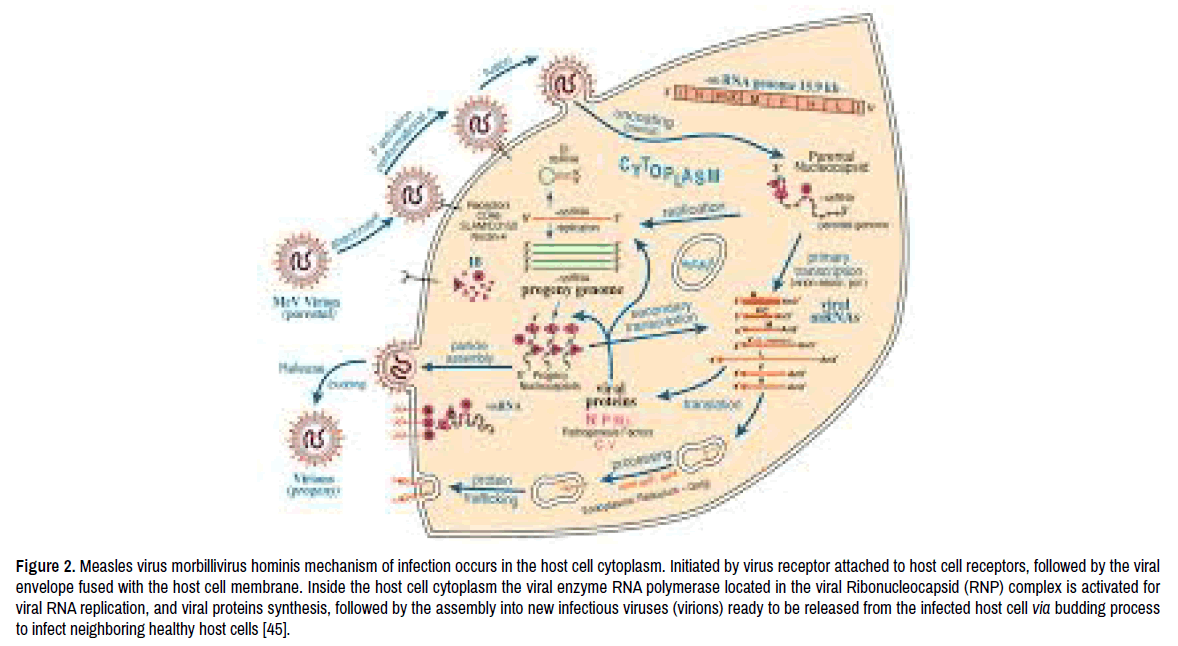
The infection with measles virus started by the virus interring the host respiratory tract, and binds to host cells receptors CD150 (SLAMF1) and (nectin-4) expressed on the surface of host immune cells of macrophages, and dendritic cells respectively, both cells are located in the host respiration system [5]. The infected macrophages and dendritic cells carries the virus into the nearby lymph nodes and into other lymph tissues where the virus replicates [6]. Virus replication started by hijacking the host cell machinery for its own replication in order to release new replicated mature infectious viruses (virions) from infected host epithelial cells, and infect other host epithelial cells causing measles illness. Measles virus replication occurred inside the host cell cytoplasm [7], and it is complicated process (Figure 2) started by the virus entry into host immune cells by the assistance of the virus outer layer two surface glycoproteins Hemagglutinin (H) and Fusion (F), both capable to attach to host specific receptors.
Figure 2. Measles virus morbillivirus hominis mechanism of infection occurs in the host cell cytoplasm. Initiated by virus receptor attached to host cell receptors, followed by the viral envelope fused with the host cell membrane. Inside the host cell cytoplasm the viral enzyme RNA polymerase located in the viral Ribonucleocapsid (RNP) complex is activated for viral RNA replication, and viral proteins synthesis, followed by the assembly into new infectious viruses (virions) ready to be released from the infected host cell via budding process to infect neighboring healthy host cells [45].
These host specific receptors are CD150 (SLAMF1) expressed on host immune cells, and Nectin-4 expressed on epithelial cells. This binding mechanism of the virus to these two host cell receptors triggers the fusion of the viral envelope with the host cell plasma membrane resulting in releasing the virus core Ribonucleocapsid (RNP) that carry the virus negative-sense RNA into the host cell cytoplasm for virus replication [8]. Inside the host cell cytoplasm, the virus RNA-dependent enzyme RNA polymerase (RdRp) composed of Phosphoprotein (P) and Large (L) proteins start to transcribe virus messenger RNAs (mRNAs) for the biosynthesis of viral proteins (N, P, M, F, H, L), and the biosynthesis of the full-length positive-sense antigenomic RNA, which serve as a template to produce newly virus negative-sense genomic RNA [9]. This machinery for the replication of the virus negative-sense genomic RNA occurs in the membrane-bound inclusion bodies located in the host cell cytoplasm. The newly synthesized virus negative-sense genomic RNA is encapsulated by the biosynthesized virus Nucleoprotein (N) to form helical nucleocapsids, and the biosynthesized virus matrix (M) protein that coordinates the virus assembly inside the host cell by bringing the newly biosynthesized virus nucleocapsids to the host cell membrane where join other biosynthesized host virus proteins for the assembly of infectious newly viruses (virions) to be released from the infected host cell via budding process [10]. This budding process leading into the destruction of infected host cell. The newly assembled and released infectious viruses (virions) infect new host healthy immune cells via the virus fusion (F) protein. Infected new immune cells of macrophages or dendritic cells carry replicated infectious new viruses (virions) to other parts of the body as secondary viremia, spreading the infection to epithelial layers, leading into to the characteristic measles symptoms of rash and respiratory making this measles virus (MeV) a contagious disease [11].
Host immune response to Measles Virus (MeV) infection
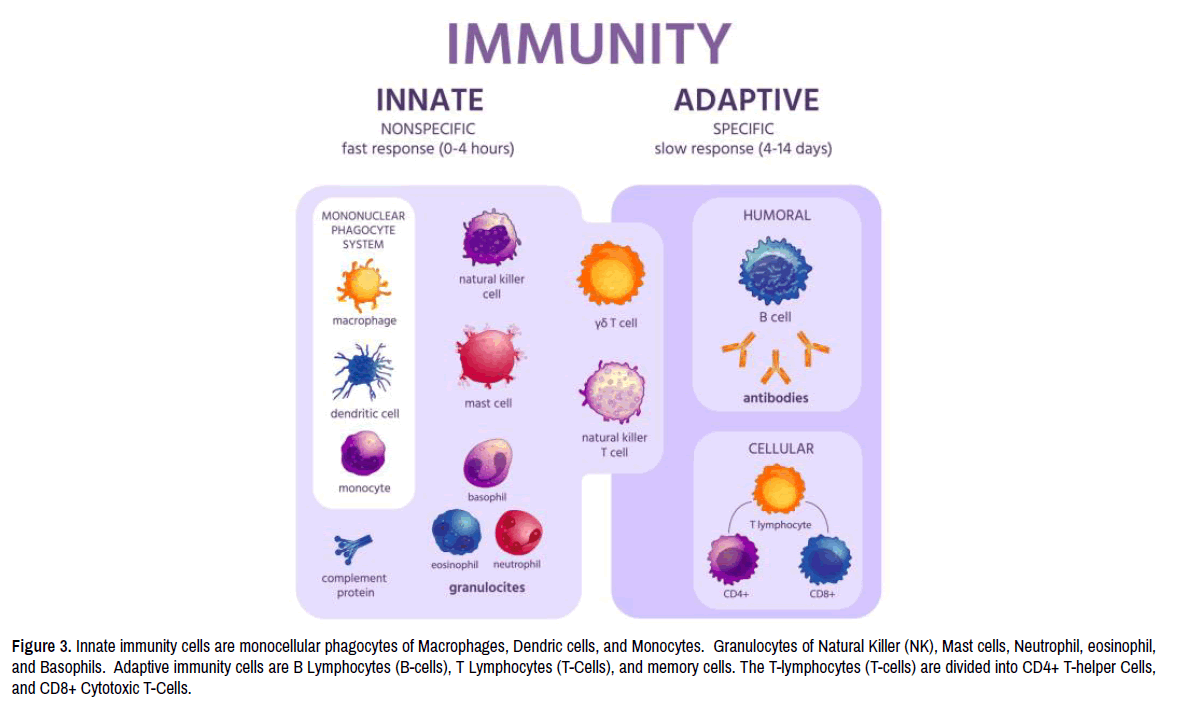
Initiation of measles virus infection to host immune cells trigger immune response to neutralize the infection for patient clinical recovery, and for the patient to build immunity against future infection with the same measles virus strain. The early host immune response is the nonspecific innate immunity [12] that normally triggered as soon the infection initiated with no symptoms occurred (dormant stage). These innate immunity cells (Figure 3) includes Natural Killer (NK) cells, a large lymphocyte that secretes enzymes like perforin to destroy infected virus structure, and also secrete cytokines like Interferon-gamma (IFN-γ) to activate specific immune response in the adaptive immunity [13]. This innate immune response is usually triggered before the host triggers adaptive immune response to measles virus infection and is capable to control the virus replication inside host cells.
Figure 3. Innate immunity cells are monocellular phagocytes of Macrophages, Dendric cells, and Monocytes. Granulocytes of Natural Killer (NK), Mast cells, Neutrophil, eosinophil, and Basophils. Adaptive immunity cells are B Lymphocytes (B-cells), T Lymphocytes (T-Cells), and memory cells. The T-lymphocytes (T-cells) are divided into CD4+ T-helper Cells, and CD8+ Cytotoxic T-Cells.
Adaptive immunity definition is the specific immune response triggered when the defense of innate immunity is failed. In the adaptive immunity antigens from the replicated measles virus is detected inside infected cells for host Antigen Presented Cells (APCs) of macrophage or dendritic immune cells capable to activate specific T and B lymphocytes T- cells, and B- cells to trigger humoral immune response and produce specific antibodies against measles virus extracellular structure proteins to inhibit the measles virus attachment to host cells receptors, and defuse into immune cells [14]. The first specific antibody produced after measles virus infect host cells are antibody IgM subtypes, followed by antibodies switch into predominantly antibody IgG3, then into antibodies IgG1 and IgG4 isotypes [15]. In addition to antibodies switch the secretory antibody IgA against the infection measles virus is secreted as mucosal secretions usually found in the host serum, and saliva in the acute measle virus infection.
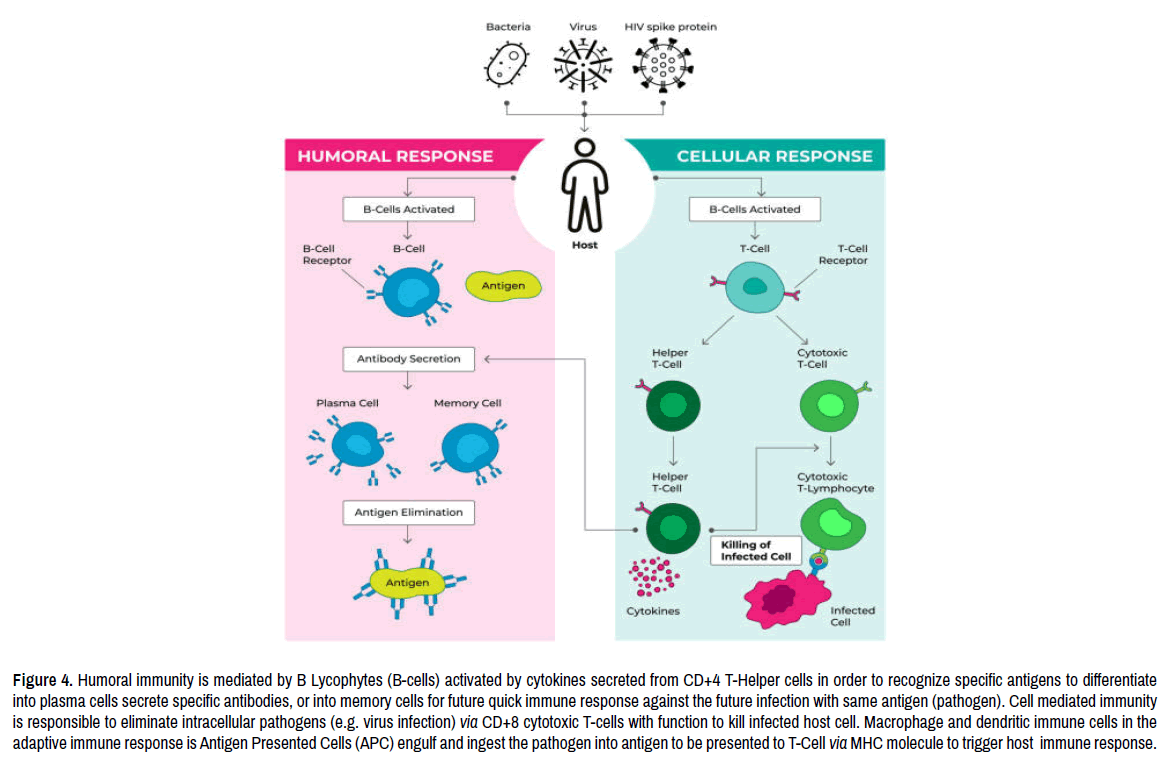
These antibodies are mainly against the virus Nucleoprotein (N), or against the two virus Hemagglutinin (H) and Fusion (F) glycoproteins located on the surface of measles virus cell. These specific antibody’s function is to neutralize the virus by inhibiting the virus internalization and replication. It is important to highlight that Antigen Presenting Cells (APCs) like dendritic cells and macrophages are linked to both innate immunity, and adaptive immunity. In the case of innate immunity both dendritic, and macrophages cells play crucial function in engulfing infected measles virus to protect host cells from the infection, but in the case of adaptive immunity both dendritic and macrophages cell’s function are Antigen Presented Cells (APCs) presenting measles virus antigens to T- cells for the initiation of the adaptive immune response for both humoral and cellular immunity (Figure 4). It is also important to highlight that there are two types of T lymphocytes (T-cells). These T-cells are T-helper cells (CD4+), and cytotoxic T-cells (CD8+). Both are critical for neutralizing measles virus infection. T-helper (CD4+) cells support B -cells in adaptive immunity for specific antibody secretion, while cytotoxic T- cells (CD8+) are responsible for killing host cells infected by measles virus in order to reduce viral load in host immune cells [16].
Figure 4. Humoral immunity is mediated by B Lycophytes (B-cells) activated by cytokines secreted from CD+4 T-Helper cells in order to recognize specific antigens to differentiate into plasma cells secrete specific antibodies, or into memory cells for future quick immune response against the future infection with same antigen (pathogen). Cell mediated immunity is responsible to eliminate intracellular pathogens (e.g. virus infection) via CD+8 cytotoxic T-cells with function to kill infected host cell. Macrophage and dendritic immune cells in the adaptive immune response is Antigen Presented Cells (APC) engulf and ingest the pathogen into antigen to be presented to T-Cell via MHC molecule to trigger host immune response.
Common symptoms after the infection with Measles Virus (MeV)
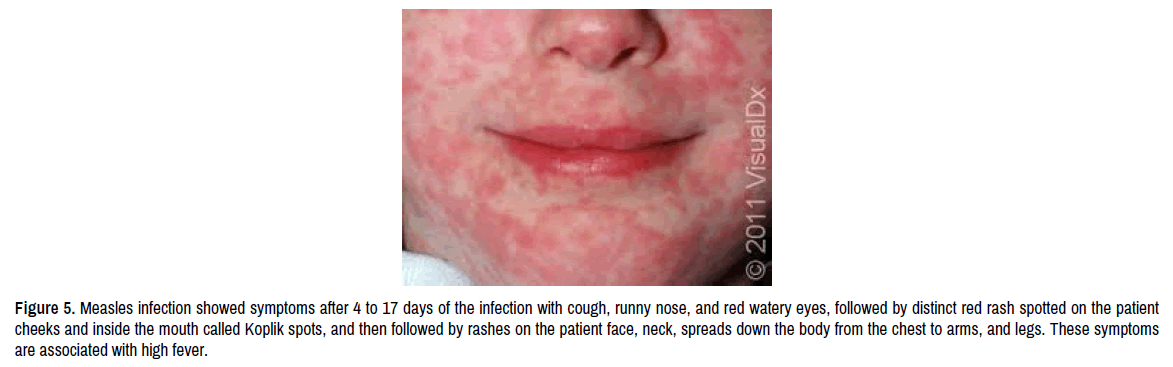
Incubation period after the person infected with measles virus is from 7 to 14 days. The first symptoms are usually fever, dry cough, runny nose, and conjunctivitis (watery red eyes), after 2 to 3 days from this first symptoms, a tiny bluish-white spots with red halos are spotted on the patient cheeks and inside the mouth called Koplik spots. This appeared Koplik spots is followed by rashes on the patient face, neck and spreads down the body from the chest to arms, and legs (Figure 5). As soon these rashes appeared the patient temperature spike for more than 104-degree Fahrenheit (400 ℃). These rashes are made of large flat spots and small raised bumps are a key diagnostic sign of measles virus infection. It is important to highlight that measles virus infection is highly contagiousness disease, and infected patient can spread the virus before the rash symptom appears.
Figure 5. Measles infection showed symptoms after 4 to 17 days of the infection with cough, runny nose, and red watery eyes, followed by distinct red rash spotted on the patient cheeks and inside the mouth called Koplik spots, and then followed by rashes on the patient face, neck, spreads down the body from the chest to arms, and legs. These symptoms are associated with high fever.
A non-vaccinated person is highly suspectable for the infection comparing to vaccinated person this is an indication of the important of vaccination for the protection from measles virus infection. Because measles virus infection is highly contagious a person with measles symptoms should stay home to prevent spreading this disease into healthy population. Measles symptoms usually take its course for recovery, and the only treatment is focusing on normal support care to ensure the patient is comfortable in order to avoid any symptom’s complications. In the case of children younger than 5 years, Pregnant women, and people with weakened immune systems are more likely to suffer more serious complications after measles virus infection such as Pneumonia a second bacterial infection, and encephalomyelitis a symptom involve central nervous system [17]. Also, few measles patients with weakened immune systems might suffer a late long-term complication such as rare progressive neurological disease known by the name Subacute Sclerosing Panencephalitis (SSPE) resulted from acquired measles virus infection earlier in life [18].
Diagnostic methods for Measles Virus (MeV) infection
Measles illness diagnostics started first by recognizing common classic symptoms after the infection which are high fever, cough, runny nose, red eyes, and spreading rashes specially the tiny white spots (Koplik's spots) inside the mouth. Laboratory tests for diagnostic confirmation are based on the detection of measles virus RNA or responded antibodies in patient specimen like throat, nose, blood, or urine. Laboratory test methods are mainly Real-time Reverse Transcription-Polymerase Chain reaction (rRT-PCR) for virus genome detection [19], and the serology test for IgM antibodies detection in the patient blood (serum or plasma) against early measles virus infection [20].
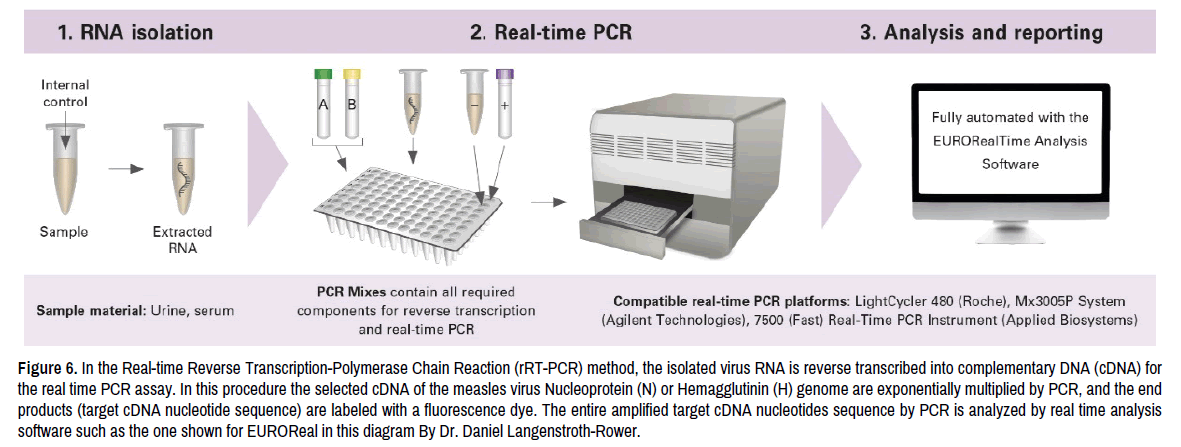
Real-time Reverse Transcription-Polymerase Chain reaction (rRT-PCR) is the preferred method for virus RNA detection from patient throat, nasopharyngeal swab, or urine samples [21]. This rRT-PCR, is highly sensitive method for measles virus RNA detection with over 94% accuracy. This method uses synthetic specific primer, and nucleotide sequence of virus nucleoprotein (N) or Hemagglutinin (H) as a template for amplification, and for the virus detection in patient specimen. The method (Figure 6) can be summarized into the following: Sample collection from the patient, viral RNA extraction from patient specimen using the commercial test kits, use test kit prepared master mix to mix the extracted virus RNA from patient specimen, the target virus nucleotide sequences of nucleoprotein (N),or Hemagglutinin (H) in the extracted virus RNA is used as a template in the PCR procedure, the specific synthetized primers (probes) sequence is used in PCR for target (template) virus nucleotide sequence amplification, the two enzymes reverse transcriptase, and DNA polymerase are used for target virus RNA reverse transcribed into cDNA for replication, finally after the replication is completed the enzyme inhibitor Ribonuclease (RNase) [22] is added to stop PCR replication reaction. This typical rRT-PCR procedure involves the following parameter: 500C for 20 minutes to convert virus RNA into cDNA by reverse transcriptase enzyme, 950 C for 2 to 15 minutes for virus cDNA denaturation, in the PCR thermocouple instrument cycling (40-45 cycles). Each cycle is 15-20 seconds for double strand cDNA denaturation, one minute for synthetic primer annealing, and for the target nucleotide sequence extension by DNA polymerase. After the procedure is completed (usually takes about 2 hours for the virus cDNA amplification), Results can be analyzed by interpreting fluorescence signals to identify the presence of measles virus nucleoprotein (N) or hemagglutinin (H) cDNA amplified plot on a semi-log plot in order to analyze amplification efficiency. The purpose of plotting is to determine the Cyclic Threshold (CT) value which is used to calculate the initial concentration of started amount of cDNA for the virus target nucleoprotein (N), or Hemagglutinin (H) glycoproteins genes in patient specimen. The sample is considered positive if the FAM fluorescence (Carboxy-fluorescein) is used as a signal in the test. The Crosses Threshold (CT) value should be able to detected positive results before the 40-45 cycles is completed in the rRT-PCR procedure.
Figure 6. In the Real-time Reverse Transcription-Polymerase Chain Reaction (rRT-PCR) method, the isolated virus RNA is reverse transcribed into complementary DNA (cDNA) for the real time PCR assay. In this procedure the selected cDNA of the measles virus Nucleoprotein (N) or Hemagglutinin (H) genome are exponentially multiplied by PCR, and the end products (target cDNA nucleotide sequence) are labeled with a fluorescence dye. The entire amplified target cDNA nucleotides sequence by PCR is analyzed by real time analysis software such as the one shown for EUROReal in this diagram By Dr. Daniel Langenstroth-Rower.
Serology test method by using Enzyme-Linked Immunosorbent Assay (ELISA) is sensitive and presumptive method used to detect IgM antibodies for acute measles infection, and also, used to detect IgG antibodies for immunity/vaccination status in tested patient blood serum or plasma. These two IgM, and IgG detection methods use 96-well plates coated with measles anti-human IgM or anti-human IgG for the detection IgM, or IgG respectively. The 96-will plates are coated with anti-IgM for IgM detection in patient blood serum or plasma provides fast presumptive diagnosis results. For IgM test method patient blood is collected within the first few days of appeared rashes before patient IgM antibodies switched into IgG antibodies [23]. Such antibodies switching in patient blood could cause false negative result in IgM detection method. Commercial Enzyme Linked Immunoassays (ELISA) test kits for IgM antibodies against measles virus infection are available in the market.
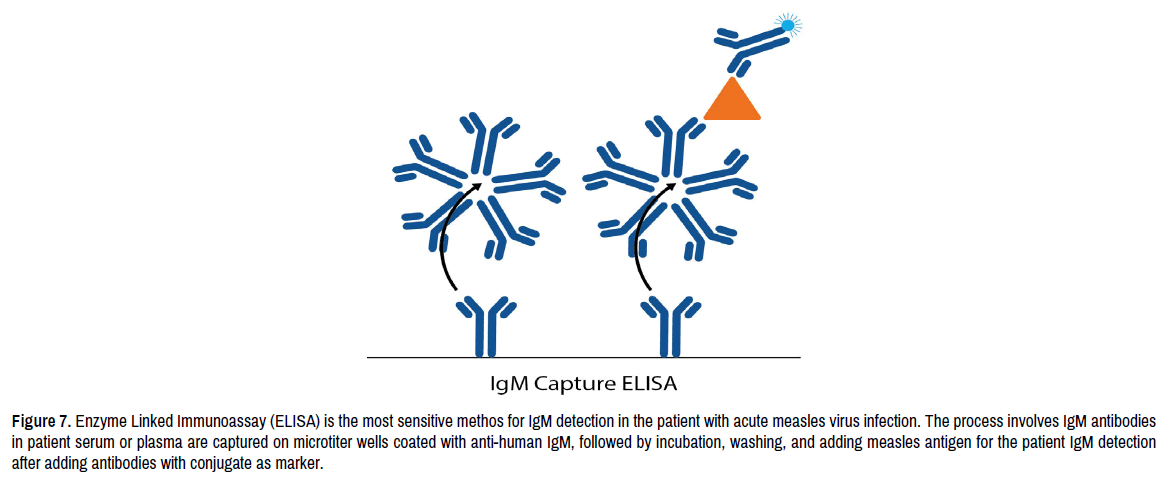
The test method in summary (Figure 7) is initiated by the separation of serum or plasma from the collected patient blood sample, Serum or plasma is diluted using buffer solution, microplate wells coated with anti-human IgM is used for capturing patient IgM antibodies in the serum or plasma, diluted patient serums or plasma is added into microplate wells (If IgM antibody specific for measles infection is presented in the patient serum or plasma, it will captured by the coated anti-human IgM in the well). After the recommended incubation period, the plate is washed with buffer solution for about five wash cycles to remove unbound patient serum or plasma components. For the IgM detection the virus antigens (often recombinant nucleocapsid protein), and enzyme Peroxidase labeled- antibody specific to the measles antigen is added for the conjugated patient IgM captured by the coated anti-human IgM in the plate well, the peroxidase enzyme substrate Tetramethylbenzidine (TMB) is added to the well to develop a color proportional to the amount of IgM in patient serum or plasma (if the IgM is present).
Figure 7. Enzyme Linked Immunoassay (ELISA) is the most sensitive methos for IgM detection in the patient with acute measles virus infection. The process involves IgM antibodies in patient serum or plasma are captured on microtiter wells coated with anti-human IgM, followed by incubation, washing, and adding measles antigen for the patient IgM detection after adding antibodies with conjugate as marker.
Diluted sulfuric acid is added to stop the enzymatic reaction with the substrate, finally, test result can be interpreted by reading the Optical Density (OD) at 450 nm using spectrophotometer. No color developed in this Enzyme-Linked Immune Assay (ELISA) test indicated negative results, meaning IgM antibodies is not in the patient serum or plasma, and the patient is presumptively not infected by the measles virus. The calculation of the IgM concentration in the sample is by plotting standard curve, and the concentration of IgM is proportional to the OD450 color value reading. Because the limitation in this Enzyme-Linked Immunoassays (ELISA) test method. It is suggested ELISA test results should be confirmed by rRT-PCR [24] if possible.
Treatments after the Measles Virus (MeV) infection
Currently, there is no specific antiviral treatment for measles. Measles virus infection has to run its course, and patients should drink plenty of liquids, take vitamin A, get lots of rest, and stay home to prevent spreading the virus to other people. However, in the case of immunocompromised persons the nonspecific antiviral ribavirin or immunoglobulin can be used for treatment [25, 26]. Also, in the case of a patient suffered from secondary bacterial infection which is the major death after measles virus infection antibiotics like Amoxicillin can be used for treatment or for the prevention from such second bacterial infection [27]. The most common secondary bacterial infection is usually by Streptococcus pneumoniae, or Haemophilus influenza. Both are the common cause of bacterial pneumonia, otitis media, skin infection, eye infection, and severe mouth ulcers [28]. Also, for nonspecific treatment it is recommended nutritional support like vitamin A is given for measles symptoms managements. World Health Organization (WHO) recommended 200,000 IU of vitamin A twice a day for 12 months of age or older children, and 100,000 IU vitamin A for children less than 12 months of age showing measles illness symptoms [29].
Vaccination for the prevention from Measles Virus (MeV) infection
There are two types of vaccines currently are used to protect healthy person from measles virus infection. These are MMR vaccine, and MMRV vaccine. Both are alive attenuated viruses with combination of other whole viruses’ antigens as vaccines. MMR vaccine contains a combination of attenuated live viruses of measles, mumps, and rubella to protect people from infectious measles, mumps, rubella viruses [30]. The immunity from this vaccine against measles demonstrated to decreases by over time. The second MMRV vaccine contains a combination of attenuated live viruses of measles, mumps, rubella, and varicella (chickenpox) to protect people from infectious measles, mumps, rubella, and varicella viruses [31]. United States Food and Drug Administration (FDA) approved MMRV vaccine for injection as a way to prevent or reduce measles virus infection, and from the severity of measles symptoms.
This MMRV vaccine is also used in United States to protect from other three infectious viruses. MMRV vaccine for the protection from infectious measles virus recommended to be administered subcutaneous injection within 72 hours of exposure to measles virus, and is recommended to be administered for children age from 12 months to 12 years in two doses (0.5 ml/dose). The first injection dose is given to toddlers at the age of 12–15 months, and the second injection dose is given to children at the age of 4–6 years [32]. This MMRV vaccine has miner side effect with rare high risk of fever associated with seizures especially for toddles when the vaccine is administered in one injection not in two separate injections as recommended [33]. MMRV vaccine is also recommended for adults, and generally is safe for both children and adult if administered as recommended. It is important to highlight that MMRV vaccine should not administered to children with severe allergies especially if the vaccine containing gelatin as expedient or neomycin as antibiotic [34]. It is important to highlight that attenuated live viruses as vaccines are generally sensitive to light, and lose its efficacy if stayed at 370 C for longer than one hour, plus it should store in the freezer with no light to maintain its efficacy.
Discussion
Measles disease was eliminated from United State in the year 2000, but in the year 2025 a massive outbreak occurred, and continue for the year 2026. Most of these cases are for children younger than 5 years old of age. These comeback measles cases with high hospitalization rate, and few deaths caused a great concern, with urgent attention on measles virus research focusing on the virus mechanism of infection and replication inside host cells in order to develop specific vaccines with higher rate of immune response, and to develop specific antiviral drugs that is currently lacking worldwide for the treatment from measles infection. The current used MMRV vaccine in United State that is mainly licensed to protect children from 1 to 12 years of age from measles virus infection has a rare serious side effect of Febrile seizure to toddlers in the age from 12 to 23 months specially if the vaccination did not administer to toddlers as preferred by CDC (administer MMR vaccine and Varicella vaccine in a separate injection). This in addition the two available MMR and MMRV vaccines used for immunization are old technology of the attenuation of isolated wild-type measles virus, mixed with other attenuated viruses in one vaccine. This technology of attenuated live viruses as vaccine was licensed in the year 1963, and are manufactured based on in vitro passage the isolated wild-type viruses in chicken embryo cells multiple time to weaken the virus pathogenicity. These attenuated live viruses as vaccine is not suitable for the vaccination of toddlers under 6-month of age, pregnant women, and immunocompromised patients.
Recently, there are new advanced technologies for vaccines manufacturing that are promising in developing specific measles vaccines with higher efficacy, stability and safety for all ages including infants, pregnant women, and immunocompromised patients. These new advanced technologies are peptide-based subunit vaccines, and DNA-based vaccines. The peptide-based subunit vaccine can be developed by manufacturing synthetic peptide sequences for measles virus cell surface glycoproteins of Hemagglutinin (H) or Fusion (F) as a vaccine, in order to trigger the host immune response with specific antibodies against the virus surface glycoproteins of Hemagglutinin (H) or Fusion (F) responsible for the virus attachments to host immune cell receptor CD150 (SLAMF1), or to endothelial cell receptor Nectin-4 for the virus internalization into host cell cytoplasm to replicate. Such vaccine can also be administered as nasal spray injection to block the virus infection progress in replacement to the traditional subcutaneous injection [35].
The DNA-based vaccine can be developed based on the concept of DNA-plasmid construction for immunization via subcutaneous injection [36, 37]. This technology is based on genetically engineered plasmid capable to express measures virus fusion glycoproteins (F) via transcription, and translation inside the host cell after subcutaneous injection. The expressed virus Fusion (F) glycoprotein inside host cell will trigger host immune response with specific antibodies against the virus Fusion (F) glycoproteins capable to block the attachment of infectious virus Fusion (F) glycoprotein to host immune cell receptor endothelial cell receptor Nectin-4 in order for the virus internalize into host cells cytoplasm for replication. This technology of DNA-based vaccine such as plasmid or nucleotide sequence of mRNA for immunization demonstrated to be successful approach for the world protection from Coronavirus infection that caused (Covid-19) pandemic in the year 2019, and 2020. The developed mRNA vaccine for the protection from Coronavirus infection (Covid-19) demonstrated to be saucerful in protecting people globally from this deadly Covid-19 illness [38].
Initiating research on the above two advanced technologies for the vaccination against measles virus infection might eradicate future measles outbreaks, plus might eliminate adverse events occurred from these attenuated live viruses as vaccines. This in addition, the possibility in stimulating long-lasting cellular and humoral immunity to protect infants under 6 months old from measles virus infection, capable to bypass maternal antibody interference [39]. This in addition it, might provide safe protection for pregnant women, and immunocompromised patients by reducing immunosuppression [40].
Developing specific antiviral drug for the treatment from this highly contagious measles virus infection is very important globally specially for infants, pregnant women, and immunocompromised patients who are unable to receive live-attenuated vaccines for immunization due to their weak immunity. Currently there is no specific antiviral drug available globally for measles infection treatment even there is high risks of long-term late complications of Subacute Sclerosing Panencephalitis (SSPE) symptoms that might occur later after measles virus infection. Developing antivirals drugs for the treatment from measles virus infection need understanding the virus mechanism of infection in order to disrupt virus internalization, and replication inside host cells to terminate the disease symptoms. It is important to highlight that measles virus mechanism of infection inside host cells are well understood for utilization in developing specific antiviral drug for the treatment after measles virus infection. Potential developing of such antiviral drugs can be by chemical or biological means with mechanism to Interfere with virus Fusion (F) glycoproteins attachment to host cell receptors Nectin-4 [41, 42] for internalization inside host cells and replication.
Antiviral drug with such mechanism will stop the virus replication inside host cell, and the patient can be recovered in short time from measles illness and symptoms. Other suggested mechanism in developing antiviral drug is to block the virus RNA replication, and assemble inside the host cell cytoplasm in order to block the release new infectious mature viruses (virion) capable to infect the host neighboring cells for the continuation of measles virus infection, and illness symptoms. Such mechanism can be with a function to inhibit the activity of the enzyme RNA-dependent RNA polymerase (RdRp) complex in order to block measles virus RNA replication inside host cell, or blocked for newly virus assembly. Such suggested mechanism will terminate releasing (budding) newly infectious mature viruses (virion) from infect host cell to infect neighboring host health cells [43]. These suggested mechanisms were successfully used in developing antiviral drug for the treatment from Coronavirus infection causing Covid19 disease [44]. Using these technologies in developing antiviral drugs for the treatment from measles virus infection and illness symptoms will quickly reduce virus load in patient cells, leading into quick recovery, and in the same time stop this contagious virus from infecting other healthy people causing outbreaks [45].
Conclusion
Measles virus infection is highly contagious mostly in winter and spring, spread from infected individual to healthy individuals. In order to avoid global measles outbreaks, the developing of specific vaccine is the only effective defense for the protection from this virus infection, and developing specific antiviral drugs for the treatment is very important in order to stop future outbreaks.
Acknowledgement
None.
Conflict of Interest
None.
References
- Malvoisin, Etienne and T. Fabian Wild. "Measles virus glycoproteins: Studies on the structure and interaction of the haemagglutinin and fusion proteins." J Gen Virol 74 (1993): 2365-2372.
- Guryanov, Sergey G., Lassi Liljeroos, Prasad Kasaragod and Tommi Kajander, et al. "Crystal structure of the measles virus nucleoprotein core in complex with an N-terminal region of phosphoprotein." J Virol 90 (2016): 2849-2857.
- Wang, Dong, Ge Yang and Bin Liu. "Structure of the measles virus ternary polymerase complex." Nat Commun 16 (2025): 3819.
- Patterson, John B., Diane Thomas, Hanna Lewicki and Martin A. Billeter, et al. "V and C proteins of measles virus function as virulence factors in vivo." Virol 267 (2000): 80-89.
- Gonçalves-Carneiro, Daniel, Jane A. McKeating, and Dalan Bailey. The measles virus receptor SLAMF1 can mediate particle endocytosis. J Virol 91: (2017) 10-1128.
- Welstead, G. Grant, Caterina Iorio, Ryan Draker and Jane Bayani, et al. "Measles virus replication in lymphatic cells and organs of CD150 (SLAM) transgenic mice." Proc Natl Acad Sci102 (2005): 16415-16420.
- Zhou, Yuqin, Justin M. Su, Charles E. Samuel and Dzwokai Ma. "Measles virus forms inclusion bodies with properties of liquid organelles." J Virol 93 (2019): 10-1128.
- Plumet, Sébastien, W. Paul Duprex and Denis Gerlier. "Dynamics of viral RNA synthesis during measles virus infection." J Virol 79 (2005): 6900-6908.
- Castaneda, Sharon J. and Timothy C. Wong. "Measles virus synthesizes both leaderless and leader-containing polyadenylated RNAs in vivo." J Virol 63 (1989): 2977-2986.
- Manié, Serge N., Sylvain Debreyne, Séverine Vincent and Denis Gerlier. "Measles virus structural components are enriched into lipid raft microdomains: a potential cellular location for virus assembly." J Virol 74 (2000): 305-311.
- Moss, William J. and Diane E. Griffin. "What’s going on with measles?." J Virol 98 (2024): e00758-24.
- Griffin, Diane E. "The immune response in measles: Virus control, clearance and protective immunity." Viruses 8 (2016): 282.
- Hammer, Quirin, Timo Rückert and Chiara Romagnani. "Natural killer cell specificity for viral infections." Nat Immunol 19 (2018): 800-808.
- Fujinami, Robert S., Xinmin Sun, Joseph M. Howell and James C. Jenkin, et al. "Modulation of immune system function by measles virus infection: Role of soluble factor and direct infection." J Virol 72 (1998): 9421-9427.
- Isa, María Beatríz, Laura Martínez, Miguel Giordano and Carlos Passeggi, et al. "Comparison of immunoglobulin G subclass profiles induced by measles virus in vaccinated and naturally infected individuals." Clin Vaccine Immunol 9 (2002): 693-697.
- Albarbar, Balid. "Human immune responses to measles virus: A literature review." J Microbiol Infect Dis 14 (2024): 103-103.
- Jain, Reena and Roosy Aulakh. "Measles-associated CNS complications: A review." J Child Sci 12 (2022): e172-e181.
- Jafri, Sidra K., Raman Kumar and Shahnaz H. Ibrahim. "Subacute sclerosing panencephalitis–current perspectives." Pediatr Health Med Ther (2018): 67-71.
- Broadhurst, M. Jana, Natasha Garamani, Zoe Hahn and Becky Jiang, et al. "Evaluation of a measles virus multiplex, triple-target real-time RT-PCR in three specimen matrices at a US academic medical center." J Clin Virol 136 (2021): 104757.
- Hiebert, Joanne, Vanessa Zubach, Carmen L. Charlton and Jayne Fenton, et al. "Evaluation of diagnostic accuracy of eight commercial assays for the detection of measles virus-specific IgM antibodies." J Clin Microbiol 59 (2021): 10-1128.
- Shimizu, H., C. A. McCarthy, M. F. Smaron and J. C. Burns. "Polymerase chain reaction for detection of measles virus in clinical samples." J Clin Microbiol 3 (1993): 1034-1039.
- Hummel, Kimberly B., Luis Lowe, William J. Bellini and Paul A. Rota. "Development of quantitative gene-specific real-time RT-PCR assays for the detection of measles virus in clinical specimens." J Virolog Method 132 (2006): 166-173.
- Landry, Marie Louise. "Immunoglobulin M for acute infection: True or false?." Clin Vac Immunol 23 (2016): 540-545.
- Woo, Gibson KS, Ann H. Wong, W. Y. Lee and C. S. Lau, et al. Comparison of laboratory diagnostic methods for measles infection and identification of measles virus genotypes in Hong Kong." J Med Virol 82 (2010): 1773-1781.
- Solomon, Tom, C. Anthony Hart, Sobhan Vinjamuri and Nicholas J. Beeching, et al. "Treatment of subacute sclerosing panencephalitis with interferon-α, ribavirin and inosiplex." J Child Neurol 17 (2002): 703-705.
- Liatsos, George D. "Controversies’ clarification regarding ribavirin efficacy in measles and coronaviruses: Comprehensive therapeutic approach strictly tailored to COVID-19 disease stages." World J Clin Cases 9 (2021): 5135.
- Karelitz, Samuel and Henry D. Isenberg. "The anti-biotic prophylaxis of bacterial complications of measles." (1959): 1-10.
- Slifka, Mark K., Dirk Homann, Antoinette Tishon and Robb Pagarigan, et al. "Measles virus infection results in suppression of both innate and adaptive immune responses to secondary bacterial infection." J Clin Invest 111 (2003): 805-810.
- Suchdev, Parminder S., Nancy F. Krebs and Sherry A. Tanumihardjo. "Reevaluating vitamin A for measles management in high-income settings." Lancet Reg Health Am 48 (2025).
- Altindag, Onur, Jane Greve and Erdal Tekin. "Impact of Measles, Mumps, and Rubella Vaccination on Hospitalizations and Human Capital: Evidence from Copenhagen School Health Records." Vaccines 13 (2025): 302.
- Yan, Kai, Guang Yang, Lixuan Yan and Ruopei Wu, et al. "MMRV vaccine safety: A comprehensive analysis of adverse events recorded among pediatric populations." Hum Vaccin Immunother 21 (2025): 2590875.
- Ackerson, Bradley K., Bonnie H. Li, Lina S. Sy and T. Craig Cheetham, et al. "Association of the use of MMRV in infants by pediatric infectious disease specialists with that of other affiliated providers." Vaccine 32 (2014): 1863-1868.
- Schink, Tania, Jakob Holstiege, Frank Kowalzik and Fred Zepp, et al. "Risk of febrile convulsions after MMRV vaccination in comparison to MMR or MMR+ V vaccination." Vaccine 32 (2014): 645-650.
- Pool, Vitali, M. Miles Braun, John M. Kelso and Gina Mootrey, et al. "Prevalence of anti-gelatin IgE antibodies in people with anaphylaxis after measles-mumps-rubella vaccine in the United States." Pediatrics 110 (2002): e71-e71.
- Mathieu, Cyrille, D. Huey, E. Jurgens and J. C. Welsch, et al. "Prevention of measles virus infection by intranasal delivery of fusion inhibitor peptides." J Virol 89 (2015): 1143-1155.
- Yang, K., F. Mustafa, A. Valsamakis and J. C. Santoro, et al. "Early studies on DNA-based immunizations for measles virus." Vaccine 15 (1997): 888-891.
- Webster, Diane E., Michelle L. Cooney, Zhongjun Huang and Damien R. Drew, et al. I "Successful boosting of a DNA measles immunization with an oral plant-derived measles virus vaccine." J Virol 76 (2002): 7910-7912.
- Ding, Yuan, Feng Fan, Xin Xu and Gan Zhao, et al. "A COVID-19 DNA vaccine candidate elicits broadly neutralizing antibodies against multiple SARS-CoV-2 Variants including the currently circulating Omicron BA. 5, BF. 7, BQ. 1 and XBB." Vaccines 11 (2023): 778.
- Niewiesk, Stefan. "Maternal antibodies: Clinical significance, mechanism of interference with immune responses and possible vaccination strategies." Front Immunol 5 (2014): 446.
- Ison, Michael G. "Vaccines for use in special populations: Immunocompromised hosts." J Infect Dis 231 (2025): 552-555.
- Plattet, Philippe, Lisa Alves, Michael Herren and Hector C. Aguilar. "Measles virus fusion protein: Structure, function and inhibition." Viruses 8 (2016): 112.
- Bovier, Francesca T., Ksenia Rybkina, Sudipta Biswas and Olivia Harder, et al. "Inhibition of measles viral fusion is enhanced by targeting multiple domains of the fusion protein." ACS nano 15 (2021): 12794-12803.
- White, Laura K., Jeong-Joong Yoon, Jin K. Lee and Aiming Sun, et al. "Nonnucleoside inhibitor of measles virus RNA-dependent RNA polymerase complex activity." Antimicrob Agents Chemother 51 (2007): 2293-2303.
- Gudima, Georgii, Ilya Kofiadi, Igor Shilovskiy and Dmitry Kudlay, et al. "Antiviral therapy of COVID-19." Int J Mol Sci 24 (2023): 8867.
- Su, Justin M., Maxwell Z. Wilson, Charles E. Samuel and Dzwokai Ma. "Formation and function of liquid-like viral factories in negative-sense single-stranded RNA virus infections." Viruses 131 (2021): 126.
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Google Scholar, Cross Ref, Indexed at
Author Info
1Consultant Biotechnology, 7434 Korbel, Dr.Gurnee IL, 60031, USAReceived: 24-Feb-2026, Manuscript No. vcrh-26-185528; Editor assigned: 26-Feb-2026, Pre QC No. P-185528; Reviewed: 12-Mar-2026, QC No. Q-185528; Revised: 18-Mar-2026, Rev Manuscript No. R-185528; Published: 24-Mar-2026, DOI: 10.37421/2736-657X.2026.10.341
Citation: Ibrahim, Osama O. "Measles Virus (MeV) Structure, Mechanism of Infection, Symptoms, Diagnostics, Prevention and Treatment." Virol Curr Res 10 (2026): 341.
Copyright: © 2026 Ibrahim OO. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.